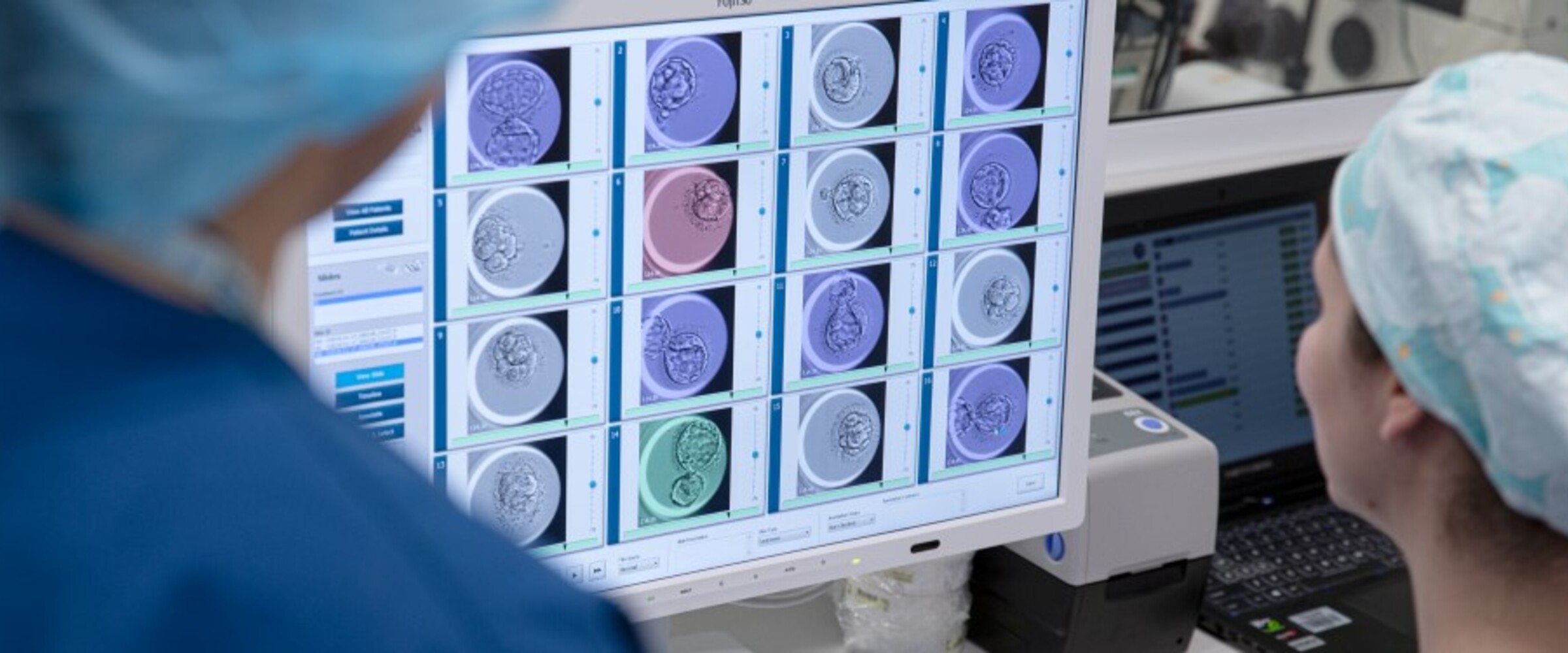
Embryoscope: the latest in early stage embryo development
Investigations into the early development of embryos using time-lapse photography are helping to predict which patterns of embryo development are associated with the best outcomes for patients.
Advances in IVF treatments over the last three decades have always been underpinned by our evolving scientific knowledge and understanding of the early stages of human development. When a new form of technology that promises to substantially increase this knowledge becomes available, it is not surprising that IVF scientists are excited about the potential implications. This is very much the case with the advent of technology which allows us to observe the undisturbed development of IVF embryos using time-lapse microscopy. Melbourne IVF and our sister unit in Sydney, IVFAustralia, are currently actively engaged in evaluating a system – the “Embryoscope” – which does just that.
For the last three decades or so, IVF scientists have gained significant insight into the very early stages of human development. These stages are able to be observed using a light microscope during the first few days following fertilisation of the oocyte (egg) by a sperm in the IVF laboratory. If we know the appropriate times to look, we can determine whether or not the fertilised egg has reached the critical checkpoints on its journey. At varying times, we can observe events such as the male and female genetic material still in separate compartments of the egg (pronuclei), the process of these pronuclei fusing (syngamy) and the egg undergoing division into two separate cells. On subsequent days we can observe further division of the embryo into smaller cells until these cells pack closely together into a structure called a morula (Latin for mulberry – I knew it would come in handy one day). The cells in the embryo then organise themselves around a fluid filled cavity that they have created to form a structure called a blastocyst.
Each time a scientist makes an observation of an embryo, it is removed from the controlled environment of the incubator and examined under a microscope before being returned as quickly as possible. Because we are very keen to minimise the number of times we disturb the embryo, we choose what we believe to be the most critical times and limit our observations to those times. We have, to date, known very little about what happens when we are not looking. The “Embryoscope” changes all that and allows us to analyse images taken at all times during development.
One of the key challenges facing IVF scientists is to predict accurately which embryo has the best chance of resulting in a pregnancy. Using our traditional observations, we have been able to show that embryos which have reached significant checkpoints in development at the time we observe them, have an increased chance of resulting in a pregnancy. Now, however, our investigation into using time-lapse images will help us to identify the exact times at which these crucial events, many of which take place in the first two days of embryo development, occur. This will allow us to better predict which patterns of embryo development are associated with the best outcomes for our patients as well as identifying those which are less likely to result in a positive outcome.
We are hopeful that the knowledge gained during this learning phase will help us to offer our patients improved prospects in the near future.
>> For more local information, visit Melbourne IVF, IVFAustralia, Queensland Fertility Group
